Boyle's Law
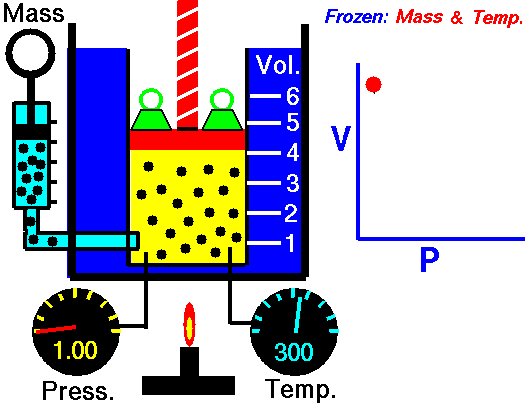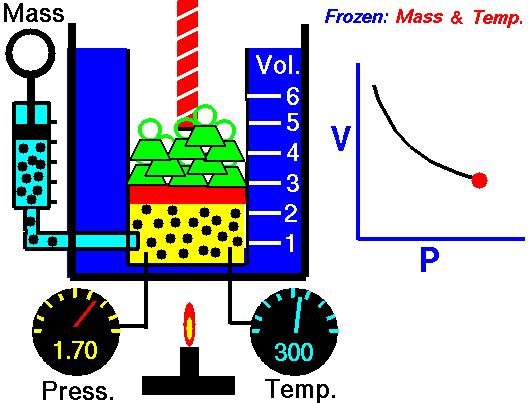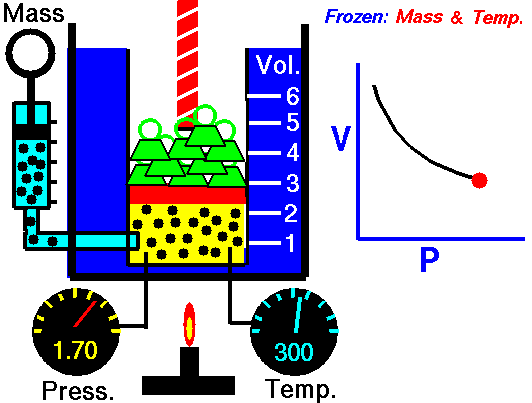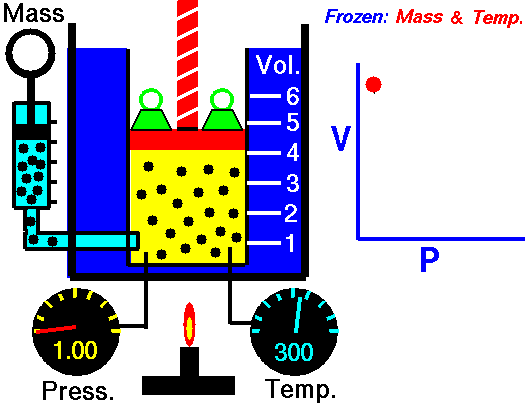
This law states that the volume of a fixed mass of gas varies inversely with the pressure at constant temperature. Simply, when the volume goes up, pressure drops, and vice versa. The mathematical equation is equally as simple: PV=K where P=Pressure, V=Volume, and K is simply a constant. This equation can also be written as P1V1=P2V2. Boyle's law is used to predict the result of introducing a change, in volume and pressure only, to the initial state of a fixed quantity of gas. Boyle found that doubling the pressure of a gas at a constant temperature reduces its volume by one-half. Similarly, if one were to triple the gas pressure, it reduces its volume to one-third of the original.
Examples of Boyle's Law
- Increase size in bubbles as they rise to the surface - The deeper we go in the sea, more the pressure. This is why as the bubbles rise to the surface the pressure decreases and the volume of the bubble increases.
- Popping a balloon - When a balloon is being squeezed, the volume is decreased, which increases the pressure. Since the balloon can't handle all of the pressure, the result is a popped balloon.
- Death of deep sea creatures due to the changes in pressure - Deep sea fish are adapted to the high pressures of the deep sea. When they are brought up to the surface the pressure inside their body is greater than the pressure of the surrounding water. This causes an imbalance and the membranes of their bladders and cells pop due to the volume increase.