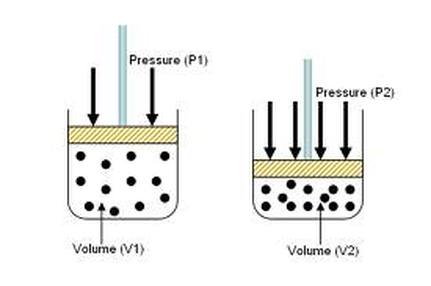
Example #1: 5.00 L of a gas is at 1.08 atm. What pressure is obtained when the volume is increased to 10.0 L?
Example #2: A gas with an unknown initial volume is at 2.45 atm pressure. Later, the same gas is measured to be at 1.60 atm pressure with a volume of 4.96 L. What is the initial volume (if the temperature is constant throughout the experiment)?